Ozempic Drives 8.5% of Canada's Public Drug Cost Growth
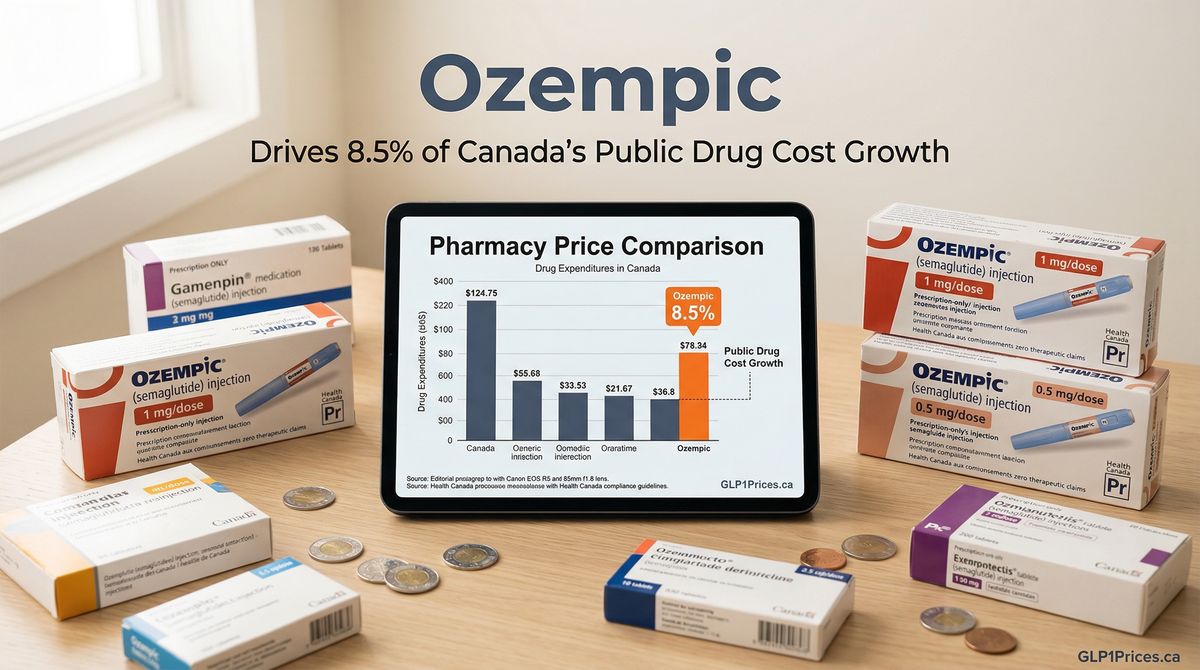
New data reveals that Ozempic has become the largest publicly funded single-drug cost driver in Canada, representing 8.5% of the total growth in public drug spending.
Public Drug Budget Impact
The finding highlights the significant financial pressure that Ozempic places on Canada's provincial drug programs and public health budgets. As a single medication, Ozempic's contribution to overall drug cost growth demonstrates the outsized impact of high-cost specialty medications on healthcare spending.
Provincial drug plans across Canada have been grappling with the rapid adoption of semaglutide-based medications, which carry monthly costs ranging from $200 to $400 per prescription depending on dosage and pharmacy.
Generic Competition Timeline
The cost pressure comes as Health Canada continues reviewing nine generic semaglutide applications following patent expiration in January 2026. The agency maintains a 180-day review timeline for generic drug submissions, with approvals potentially arriving by summer 2026.
Generic manufacturers including Sandoz, Apotex, Teva, and others have submitted applications for biosimilar versions of semaglutide. Industry analysts project generic versions could reduce monthly costs to under $100, providing significant relief to public drug budgets.
Broader GLP-1 Market Context
Beyond Ozempic, other GLP-1 medications continue entering the Canadian market. Wegovy carries an estimated annual cost of $5,000 according to Canada's Drug Agency, while Mounjaro pricing decisions remain under review by provincial plans.
The 8.5% figure represents only Ozempic's contribution to public spending growth, not including private insurance or out-of-pocket payments by Canadian patients.
Provincial Response Strategies
Provincial drug plans are implementing various strategies to manage GLP-1 costs while maintaining patient access:
- Prior authorization requirements for new prescriptions
- Step therapy protocols requiring trial of other medications first
- Coverage criteria focusing on specific medical conditions
- Monitoring programs to track utilization patterns
Looking Ahead
As generic competition approaches, provinces are preparing coverage frameworks for biosimilar semaglutide products. The potential cost savings could free up significant budget capacity for other healthcare priorities while maintaining access to GLP-1 therapy.
Patients currently using Ozempic can track generic approval progress through our generic semaglutide tracker and explore current coverage options via our insurance coverage checker.
This article is for informational purposes only and does not constitute medical advice.
Get notified when generic prices go live
We’ll send one email the moment generic semaglutide prices are listed at Canadian pharmacies. No spam.
Get notified when generic semaglutide becomes available in Canada
Expected Q3 2026 — be the first to know